Posted December 12, 2017 in Cosmetic Surgery Articles
3 minute read
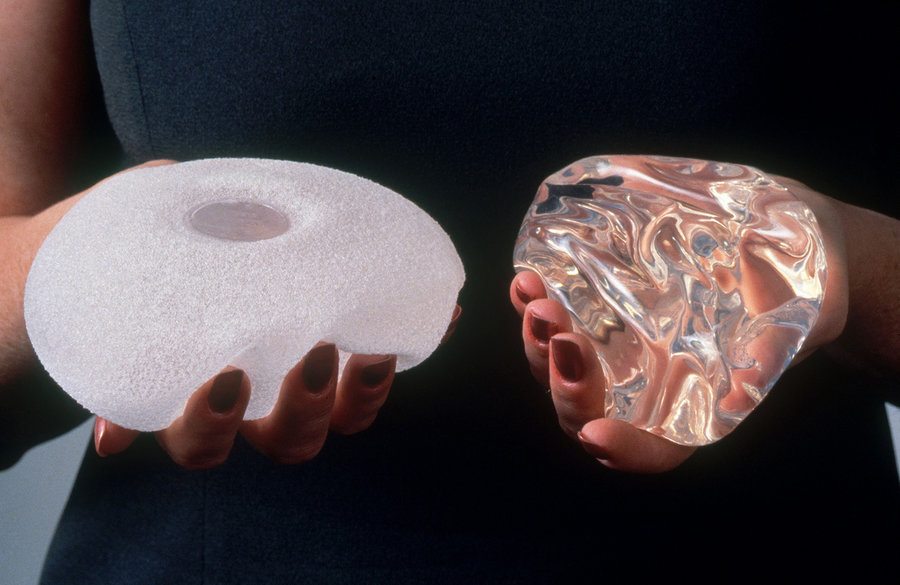
Dr. Bitar has been doing breast augmentations in private practice at the Bitar Institute for 22 years and has never used any textured implants. Furthermore, after thousands of breast implant surgeries, he has had zero cases of BIA- ALCL.
You may have heard recent media reports regarding breast implants and an uncommon form of cancer – breast implant associated anaplastic large cell lymphoma (BIA-ALCL), a form of T-cell lymphoma.
A New York Times article in May publicized an update on the topic issued by the Food and Drug Administration and has concerned many patients with implants and others considering breast augmentation surgery.
While this recent report caught the attention of the media, this is not a new entity, and the FDA first issued a report of a possible association in 2011.
What is BIA-ALCL?
BIA-ALCL is not breast cancer, but a rare and treatable form of lymphoma that develops as a fluid swelling around the implant. The FDA report in March, for the first time, offered insight as to which implant types may have a higher association with the disease.
Risk and Statistics: Understanding the Numbers
The lifetime risk appears to be about 1 case for every 30,000 women with textured implants, or a .003% risk, based on the 359 cases that have been reported world wide. Thus far, there have been no cases of BIA-ALCL in patients who have had exposure only to “smooth” implants. Note that approximately 550,000 patients get breast implants in the US each year, and it is estimated that 12.7% get textured implants. The fill of the implant, saline or silicone, seems to have no impact on the development of this condition. The lag time between augmentation and diagnosis has ranged from 2 to 28 years, and the condition has been reported in both aesthetic and reconstructive patients.
Treatment and Management of BIA-ALCL
It is important to know that simply removing the implant and surrounding scar tissue can often cure women who develop BIA-ALCL. In all but a few cases, the disease has been fully resolved by surgery alone, though rare patients may require additional treatment such as radiation or chemotherapy. Following removal, replacement with a smooth implant may even be an option.
Should Patients Have Their Implants Removed?
Currently neither the FDA or the American Society of Plastic Surgeons suggest implant removal for asymptomatic women. Current opinion is that women without breast changes do not require additional screening but should continue with routine follow up with their surgeon.
The FDA recommends, as do I, that every woman conduct regular self examination and be seen by their physician for an annual exam. If an individual develops a swelling or lump in the breast, they should be evaluated by their doctor. If they have implants, then any unexpected changes in shape or symmetry should be brought to the attention of their surgeon.
Experience Matters
Dr. Bitar, a board-certified plastic surgeon at Bitar Institute, has extensive experience in breast augmentation and other breast procedures. He prioritizes patient education and empowers women to make informed decisions about their health and well-being. If you have questions or concerns about your breast implants, Dr. Bitar is happy to discuss your individual situation and offer personalized guidance. Schedule a consultation today to learn more and address any concerns you may have.
While the information in this blog post is intended to be educational and informative, it’s crucial to remember that it cannot replace seeking professional medical advice. If you have any concerns about your breast implants or experience any changes in your breasts, it is essential to schedule a consultation with a qualified healthcare professional.
